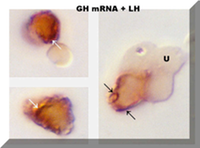
The following page describes the current non-radioactive in situ hybridization technique used by Dr. Gwen V. Childs at the University of Arkansas for Medical Sciences to detect mRNAs in whole pituitary cells grown in culture. We no longer use the avidin-biotin peroxidase complex to detect the biotinylated probes. Instead, we use a new five-step immunolabeling protocol that detects biotin conjugated to the mRNA probes with anti-biotin. The protocol can be followed by a second detection system for an antigen. The following electron micrograph shows that the protocols can be used with cells in suspension, which can then be prepared for electron microscopy. The labeling for LH mRNA can be seen on vacuoles of rough endoplasmic reticulum (RER). The micrograph in the side bar shows cells with GH antigens (orange) and LH mRNA arranged in linear patterns (arrows). Below shows labeling for GH mRNA in a circular pattern, which is likely to be on the nuclear envelope.
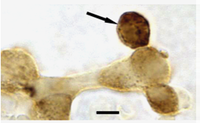
Dual in situ hybridization for GH mRNA followed by immunolabeling for GH mRNA showing a circular pattern, likely to be RER around the nuclear envelope.
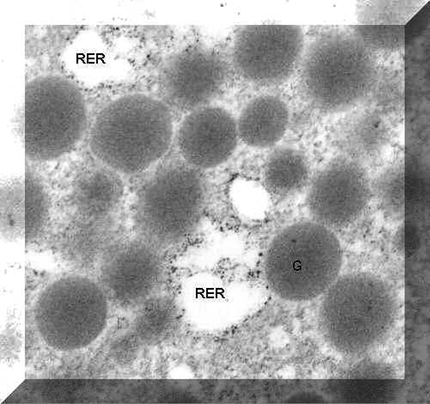
In situ hybridization protocol applied to fixed cells in suspension after which they were post fixed in osmium tetroxide and prepared for electron microscopy in a pellet. Ultrathin sections showed the label along the outside of the rough endoplasmic reticulum profiles (RER).
- Solutions
- Hybridization buffer
- Prehybridization Protocol
- Post hybridization steps: detection of hybrids
- Detection of Peroxidase
- Dual-labeling for antigens
- Illustrations of dual-labeling
- Electron micrograph illustrating labeling on rough endoplasmic reticulum.
- Publications using these methods
- Vendors for in situ hybridization histochemistry.
- Important links to courses, protocols, and services in in situ hybridization.
The protocol is applied to cells plated for 1h -36 h on 13 mm glass coverslips ( Thomas Scientific , Catalog number 6672A75) in 24 well trays ( Fisher Scientific Catalog number 08-757-156). The protocol can also be used on frozen sections or paraffin sections.
The probes used for the hybridization are either cRNA probes or complementary oligonucleotide probes at least 30 mer long. They have been biotinylated by the Vector Photobiotin kit ( Vector Laboratories,) Burlingame Calif), or with Biotin-UTP, during in vitro transcription. Most recently, they were produced by www.GeneDetect.com, with Greenstar technology. See this link for more information.
All fixation, washing, and handling methods are run under sterile conditions to prevent RNase contamination. Controls include substitution of labeled sense sequences, or omission of the labeled probe. The protocol can be adapted for use with dispersed cells at the electron microscopic level.
Solutions for in situ hybridization
- DEPC-Water (0.1%)—1ml DEPC to 999 ml H2O, stir overnight; boil the next day until 30-40% total volume is evaporated. Cool and autoclave, filter before use in all buffer solutions.
- Phosphate buffered saline
(PBS):
- 0.1 M Phosphate buffer—2.4 gm sodium phosphate (NaH2PO4), 11.36 gms sodium dibasic phosphate (Na2HPO4) in 1000 ml DEPC-H2O pH 7.4, autoclave, filter before use
- 0.1M phosphate buffer (made in DEPC water) + 0.9% NaCl pH 7.2. Autoclave and filter.
- Triton X-100 (0.3%): 300 microliters Triton X-100 ( Sigma Chemical) & bring to 100 ml.with 0.1M PBS
- 50 mM EDTA buffer : add 1.46 gm. EDTA to 100 ml. of 0.1M Tris buffer made in DEPC buffer, Catalog T-5030, (20 ml. 0.5M Tris + 80 ml. Millipore-filtered water) pH 8.0 ( Sigma Chemical)
- Diluent
for the proteinase K: 50 mM Tris with 2 mM Calcium chloride.
- Stock is 500 micrograms/ml proteinase K; add 5 micrograms of stock/1995 mls buffer for a 5 microgram/ml dilution. May go up to 10 micrograms/ml.
- 4% para-formaldehyde: 4.0 gm. p-formaldehyde in 100 ml. 0.1M PBS pH 7.2
- 0.25% acetic anhydride + 0.1M triethanolamine: 250 ul acetic anhydride & bring to 100 ml. millipore filtered water, then add 1.856 gm. triethanolamine, pH 8.0 ( Sigma Chemical)
- 20X SCC Stock—174 gms NaCl (3M), 98 gms Na citrate (0.3 M), 1000 ml DD H2), pH 7.0; aliquote and store in freezer. Dilute to make 4X, 2X and 0.1X for the post-hybridization steps.
- 50% formamide in 2X SSC: make a 1:10 dilution from 20X SSC with Millipore-filtered water and add 1:1 formamide ( Sigma Chemical), Catalog F-7503
- 4X SSC : make a 1:5 dilution from 20X SSC with millipore filtered water
- RNAse A buffer—10 mM Tris-HCl buffer (2 ml, 0.5 M Tris, 98 ml DD H20,) 1 mM EDTA—0.186 gm, 0.4 M NaCl—2.9 gms pH 8.0 filter.
Preparation
of In Situ Hybridization buffer :
The components come from Sigma
Chemical , unless otherwise noted.
- Deionize formamide: add 1.5 gm. REXYN I-300 ( Fisher Scientific, Catalog number R-208) to 100 ml. formamide, stir for 45-60 min., filter thru Whatman filter paper
- Combine 100 ml. deionized formamide + 100 ml. 4X SSC (20 ml. 20X SSC + 80 ml. millipore filtered water)
- Add 7.88 gm. Tris salt---0.25M pH 7.5 , warm to about 37 degrees in 1.0 ml. millipore filtered water
- With sterile syringe & needle add the following to the warm
formamide-Tris solution:
- 0.5 gm. bovine serum albumin (RIA grade) ( Sigma Chemical, Catalog #A-7638)
- 0.5 gm. Ficoll-400
- 0.5 gm. PVP-360
- 1.0 gm. sodium pyrophosphate
- 1.0 gm. SDS (lauryl sulfate sodium salt)
- Add 200 ul of dissolved salmon sperm DNA (ssDNA; 25 mg/ml) to 200 ml. of hybridization buffer
- Aliquot into 10 ml tubes & store frozen (-20 C) in 15 ml. centrifuge tubes
Prehybridization Steps Used for Rat or Mice Pituitary Cells Protocol
- Cells are grown on glass coverslips ( Thomas Scientific , Catalog number 6672A75), in Dulbeccos modified Eagles Medium ( JRH Biosciences , Catalog number 56499-10L), and fixed in 2.5% glutaraldehyde (in 0.1 M phosphate buffer) for 30 min. Then cells are washed for 1 h in 4 changes of phosphate buffer + 4.5% sucrose. Work under sterile conditions and store no longer than 1 wk at 2-4 C. (Note: for fluorescence labeling, cells should be fixed in 4% paraformaldehyde, 30 min).
- Rinse cells on coverlips with fresh 0.1M Phosphate buffered saline (PBS)--5 mins. room temp., shaking. Then treat in the following sequence of solutions. All of the following chemicals but the paraformaldehyde came from Sigma Chemical
- Treat with 0.3% Triton X-100---15 min. room temp.
- Wash coverslips with 0.1M PBS---2X, 3 min. each, room temp, shaking
- Treat with Proteinase K (in 50 mM Tris with 2 mM Calcium chloride.)---15 min room temp.
- Postfix with 4% p-formaldehyde/0.1M PBS---5 mins, room temp.
- Wash coverslips with 0.1M PBS---2X, 3 min. each room temp, shaking
- Treat with 0.25% acetic anhydride---10 min. room temp, shaking
- Treat with 50% formamide/2X SSC---10 min. room temp, shaking & then incubate at 37 degrees for 10 mins.
- Hybridization step: incubate cells with 200 ng/ml. of biotinylated oligonucleotide or cRNA-probe in hybridization buffer at 37 degrees overnite.
Post hybridization steps for rats or mice:
Rat Tissues
- Wash coverslips with 4X SSC---3X, 15 min. each room temp. (gentle shaking during the last 5 mins.)
- Block with 0.05M Tris buffered saline + 1% Bovine serum albumin + 10% Normal Horse Serum ( Vector Laboratories) ---15 min. room temp
- Treat with monoclonal Mouse Anti-Biotin (1:30) ( Dako, Corp )---30 min. at 37 degrees
- Wash coverslips with 0.05M Tris buffered saline---2X, 3 min. each
- Incubate with Biotinylated Horse anti-mouse IgG (rat absorbed, Vector Laboratories)1:100---10 min. room temperature
- Wash coverslips with 0.05M Tris Buffered saline---2X, 3 min. each
- Incubate with second layer of monoclonal Anti-Biotin (1:100)---30 min. at 37 degrees
- Wash coverslips with 0.05 M Tris buffered saline---2X, 3 min. each
- Incubate with second layer of Biotinylated horse anti-mouse IgG (rat absorbed) 1:100 ---10 min. room temperature
- Wash coverslips with 0.05M Tris buffered saline---2X, 3 min. each
- Incubate with 1:10 peroxidase conjugated streptavidin ( Dako, Corp, Catalog number P-397) ---5 min. Room temperature
- Wash coverslips with 0.05M Tris buffered saline—2X
- Detect peroxidase with Diaminobenzidine according to kit instructions.
Mouse Tissues
- Wash coverslips with 4X SSC---3X, 15 min. each room temp. (gentle shaking during the last 5 mins.)
- Block with 0.05M Tris buffered saline + 1% Bovine serum albumin + 10% Normal Horse Serum ( Vector Laboratories) ---15 min. room temp
- Treat with Goat Anti-Biotin (1:30) Vector Laboratories---30 min. at 37 degrees
- Wash coverslips with 0.05M Tris buffered saline---2X, 3 min. each
- Incubate with Biotinylated Horse anti-goat IgG (rat absorbed, Vector Laboratories)1:100---10 min. room temperature
- Wash coverslips with 0.05M Tris Buffered saline---2X, 3 min. each
- Incubate with second layer of Goat Anti-Biotin (1:100)---30 min. at 37 degrees
- Wash coverslips with 0.05 M Tris buffered saline---2X, 3 min. each
- Incubate with second layer of Biotinylated horse anti-Goat IgG (rat absorbed) 1:100 ---10 min. room temperature
- Wash coverslips with 0.05M Tris buffered saline---2X, 3 min. each
- Incubate with 1:10 peroxidase conjugated streptavidin ( Dako, Corp, Catalog number P-397) ---5 min. Room temperature
- Wash coverslips with 0.05M Tris buffered saline—2X
- Detect peroxidase with Diaminobenzidine according to kit instructions.
After biotinylated ligands, mRNA or a first antigen is detected we use contrasting colored substrates and immunocytochemistry to detect a pituitary antigen. This allows us to identify the cell that contains the mRNA by its antigen content. The photo in the side-bar shows the dual reaction for LH antigens (orange) and GH mRNA (arrows) in gonadotropes from proestrous rats. The following outline shows an example of the protocol that detects the second antigen (such as luteinizing hormone or follicle stimulating hormone, or growth hormone). This allow us to identify the hormone content of the cell that binds the ligand, expresses the mRNA or expresses more than one hormone. For more information, see the GH web site.
To detect antigens after the detection of the mRNA use Dual Immunolabeling:
- After in situ hybridization is completed, rinse coverslips with 0.05M Tris buffered saline---1X
- Block again with 0.05M Tris buffered saline + 1% Bovine serum albumin ( Sigma Chemical, Catalog number A-7638)---15 min. room temperature
- Incubate with either 1:30K Anti-LH/1:10K Anti-FSH---30 min. at 37 degrees
- Wash coverslips with 0.05M Tris buffered saline---3X
- Incubate with Biotinylated Goat anti-rabbit IgG ( Vector Laboratoriesl, Catalog number BA-1000) ( 25 microliters stock Biotinylated-IgG + 25 microliters Normal goat serum in 2 milliliters of buffer )---20 min., room temperature
- Wash coverslips with 0.05M Tris buffered saline---2X
- Incubate with 1:200 peroxidase conjugated streptavidin ( DAKO Corp, Catalog number P-397)---20 min. room temperature
- Wash coverslips with 0.05M Tris buffered saline---2X
- To detect peroxidase--second reaction (orange-amber), prepare orange diaminobenzidine from kit, with instructions in kit.
- Apply to cells for 5-7 min. RT
- Wash coverslips with millipore filtered water---3X
- Dehydrate, dry & permount
Warning: diaminobenzidine is eventually washed out in water soluble mounting media, especially temporary mounting media like glycerol. Therefore, if another peroxidase substrate is used, that requires water soluble media, please store the slides dry and use glycerol ONLY for short periods of time. The best way to store the slides is to dehydrate the tissues and use permount.
- Vendors for Cytochemistry Techniques
- GeneDetect for Oligoprobes. For reliable, highly sensitive anti-sense oligonucleotide probes, we use www.GeneDetect.com. This company uses a variety of labeling systems and we have had excellent success with their biotinylated and digoxygenin-labeled oligonucleotide probes for GH, LH, FSH, and leptin mRNA. They will also design the probes and provide support. Each probe comes with a kit that includes labeled sense and antisense sequences as well as unlabeled sequences for competition experiments. They respond rapidly to all requests.
- Dako Corporation, 6392 Via Real, Carpenteria, Ca 93013; 800-235-5763.
- Fisher Scientific, 10700 Rockley Road, Houston, Tx 77099; 800-876-1900 or 800-766-7000; 800-395-5442 (instrument service)
- JRH Biosciences, PO Box 14848 Lenexa, KS 66325 800-255-6032
- Sigma Chemical, PO Box 14508; St. Louis, MO 63178; 800-325-3010 Link to their Web page at Sigma Chemical Web page .
- Thomas Scientific, 99 HIgh Hill Road, PO Box 99, Suresdesboro, NJ 08085-0099, 800-345-2100 http://www.thomassci.com/
- Vector Laboratories, 30 Ingold Road, Burlingame, Ca 94010, 800-227-6666. http://www.vectorlabs.com/
For more information, contact:
Gwen Childs, Ph.D.
Professor and Chair
Department of Neurobiology and Developmental Sciences
University of Arkansas for Medical Sciences
Little Rock, AR 72205
childsgwenv@uams.edu
gvchilds@me.com