This protocol is used to detect neuropeptides and growth factors bound to target sites on pituitary cells. These ligands are first biotinylated. After tests of their potency and binding affinity, they are used on pituitary cells grown in culture for 1-5 days (time dependent on the experimental design). The cells are exposed to physiological concentrations of biotinylated ligands for 1 min to 4 h, depending on the type of experiment. After exposure, the cells are fixed for light or electron microscopic detection of the site of binding. The following figure diagrams one of the protocols we use.

The protocol described in the following paragraphs detects binding sites for biotinylated Gonadotropin releasing hormone (Bio-GnRH) at the light microscopic level. It is diagramed in the above figure. The detection system illustrated in the figure is an Avidin-Biotin-peroxidase complex solution which can be purchased from Vector Laboratories (Elite Kit). It is made 30 min before use. Avidin gold, avidin-ferritin, avidin peroxidase, or avidin fluorescein can also be used, however. If the peroxidase substrate DAB is used, it is nickel intensified and produces a purple-or gray-black reaction product.

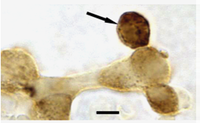
The detection protocol is then followed by immunolabeling with a contrasting color reaction product to identify one of the antigens in the target cells. Examples of cells labeled with this dual-labeling protocol are shown above, and in the side-bar. The first photo (Above) shows gonadotropes that have bound Biotinylated GnRH indicated by the arrow pointing to the patch of labeling at one cellular pole. The cells are dual labeled for GnRHR antigens (amber) showing expression of the protein as well. The top photo in the side bar shows a GH cell immunolabeled amber for GH with strong labeling for biotinylated Gonadotropin releasing hormone receptors in patches over the nucleus. Some of the patches could be in the Golgi complex area. For more information, see the GH web page
Materials:
- 0.1-10nM Biotinylated D-Lys6 GnRH or biotinylated GHRH
- 13 mm glass coverslips ( Thomas Scientific Catalog number 6672A75) on which pituitary cells are growing (1-5 day cultures) in 24 well trays ( Fisher Scientific , Catalog number 08-757-156).
- ABC Elite kit ( Vector Laboratories , Catalog number PK 6101)
- Diaminobenzidine Kit ( Vector Laboratories , Catalog number SK 4100)
- Primary antisera for hormone of interest or receptor proteins.
Method:
- Transfer coverslips into a new tray with fresh Dulbeccos Modified Eagle's media (DME) ( JRH Biosciences , Catalog number 56499-10L) + 10-4 M ascorbic acid (AA)
- Replace media once with 450 microliters/well DME + AA.
- Add 50 microlitersl/well of Bio-GnRH or Bio-GHRH for 10 mins.(24 degrees or 37 degrees C) (optimal times for maximal labeling will depend on the ligand and how fast it is taken up and/or degraded by the cells)
- Fix cells with 2% glutaraldehyde---30 mins. (room temperature (RT)
- Wash cells with 4.5% sucrose buffer + glycine---4X, 15 mins. each (RT)
- Rinse coverslips with 0.05M phosphate buffer---1X
- Block with phosphate butter containing + 5-10% normal goat serum + 0.1% bovine serum albumin---15 mins. RT
- Incubate with the avidin-biotin peroxidase complex (ABC) --30 mins. RT Prepare according to kit instructions 30 min before use.
- Wash coverslips with 0.05M phosphate buffer---1X
- Rinse coverslips with 0.05M acetate buffer—2X
- Detect peroxidase according to kit instructions using nickel intensification protocol that comes with the kit.
Warning: diaminobenzidine is eventually washed out in water soluble mounting media, especially temporary mounting media like glycerol. Therefore, if another peroxidase substrate is used, that requires water soluble media, please store the slides dry and use glycerol ONLY! for short periods of time. The best way to store them is dehydrated and mounted in permount.
This protocol can be followed by dual immunolabeling with a contrasting colored substrate (orange).
Vendors for Cytochemistry Techniques
- Dako Corporation, 6392 Via Real, Carpenteria, Ca 93013; 800-235-5763.
- Fisher Scientific, 10700 Rockley Road, Houston, Tx 77099; 800-876-1900 or 800-766-7000; 800-395-5442 (instrument service)
- JRH Biosciences, PO Box 14848 Lenexa, KS 66325 800-255-6032
- Sigma Chemical, PO Box 14508; St. Louis, MO 63178; 800-325-3010 Link to their Web page at Sigma Chemical Web page .
- Thomas Scientific, 99 HIgh Hill Road, PO Box 99, Suresdesboro, NJ 08085-0099, 800-345-2100 http://www.thomassci.com/
- Vector Laboratories, 30 Ingold Road, Burlingame, Ca 94010, 800-227-6666. http://www.vectorlabs.com/
For more information, contact:
Gwen Childs, Ph.D.
Professor and Chair
Department of Neurobiology and Developmental Sciences
University of Arkansas for Medical Sciences
Little Rock, AR 72205
childsgwenv@uams.edu
gvchilds@me.com